Boson Rapid SARS-CoV-2 Antigen Test Card
- This device is authorized by Health Canada’s under the Interim Order – Authorization #323096, so it’s in-line with the license issued for this medical device.
- Lab Based* —— For professional use
- Quantity: 20 tests/kit
- Sample type: Nasal swab
- Storage temperature: 2–30°C
- Shelf life: 18 months
- Test time: 15–20 minutes (do not read results after 20 minutes)
- Back order now, in stock by Mid-Febuary
*Lab-based tests are conducted in a lab environment by a trained laboratory technician. Point-of-care testing devices are used by an approved operator (often a health care professional) in a near-patient environment like a medical office or at the bedside. The patient waits for the results. Self-testing devices are those that can be purchased and used by the general public.
Rapid SARS-CoV-2 Antigen Test Card is a lateral flow immunoassay intended for the comparative detection of nucleocapsid protein from SARS-CoV-2 in nasopharyngeal swab specimens.
Features:
High Accuracy: nasopharyngeal swab collection with clinically proven high accuracy results
Affordability: Any qualified Health Care professionals can easily use our three-step, cost-effective portable testing kit
Easy to use: Testing can be easily administered in almost any environment without additional special equipment. Easy to transport with no special storage requirements, a test kit is all you need.
Rapid Results: Compared to standard RT-PCR tests which could take multiple days for patients to receive their results, Boson Antigen testers will show results within 15–20 minutes.
Product Information:
Quantity: 20 tests/kit
Sample type: Nasopharyngeal swab
Storage temperature: 2–30°C
Shelf life: 18 months
Test time: 15–20 minutes (do not read results after 20 minutes)
Diagnostic Sensitivity: 93.75%
Diagnostic Specificity: 98.4%
Shelf Life (expiration): 18 months
Storage Temperature: 2-30°C/36-86°F
IO authorization Device identifier : 061254/09167/1N40C5/1N40T0
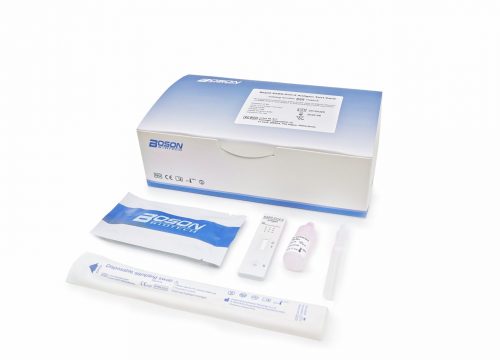
Kit Contents:
- 20 Rapid SARS-CoV-2 Antigen Test Cards (Individually packaged)
- 2 Sample Buffer
- 20 Sterilized nasopharyngeal swabs (Individually packaged)
- 20 Extraction tubes (Individually packaged)
- 1 Instruction for use
- 1 Tube rack (for 6 tubes/rack)
POC authorized use guidelines may vary by province so please contact your local regulators for confirmation on local professional use requirements.
Rapid SARS-CoV-2 Antigen Test Card is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein from SARS-CoV-2 in nasopharyngeal swab specimens from individuals who are suspected of COVID-19 by their healthcare provider within the first 7 days of symptom onset.